Three-dimensional (3D) spheroid and organoid models have become essential for preclinical drug screening because they capture gradients, cell interactions, and microarchitectures that 2D cultures cannot reproduce. But their advantages come with a challenge. Spheroids are notoriously variable. Even small differences in size, morphology, or viability can bias readouts, reduce reproducibility, and complicate comparisons across experiments.
Recent studies show how fabrication method, intrinsic cell behavior, and workflow design collectively shape spheroid uniformity and therefore the reliability of downstream drug responses. Addressing these parameters is now central to building high-throughput, high-confidence screening pipelines.
Why Uniformity Matters in 3D Spheroid Cultures for Drug Screening
Uniformity refers mainly to consistent spheroid size (diameter/volume), shape (sphericity, compactness), and internal architecture across wells and plates. When these parameters are homogeneous, spheroids display comparable diffusion gradients and cell‑cycle distributions, making responses to a given drug comparable across replicates.
Spheroid size and compactness dictate diffusion of drugs, oxygen, and nutrients, so heterogeneous diameters generate different microenvironments. Larger spheroids, starting from about 200 to 500 μm in diameter, display an external proliferating zone, an internal quiescent zone caused by limited distribution of oxygen, nutrients, and metabolites, and a necrotic core. The necrotic zone is intrinsically more drug‑resistant than the proliferative periphery, leading to size‑dependent differences in apparent potency. Irregularly shaped spheroids may contain multiple necrotic foci and heterogeneous distributions of proliferating versus quiescent cells, which significantly alter treatment response and complicate interpretation (Figure 1).
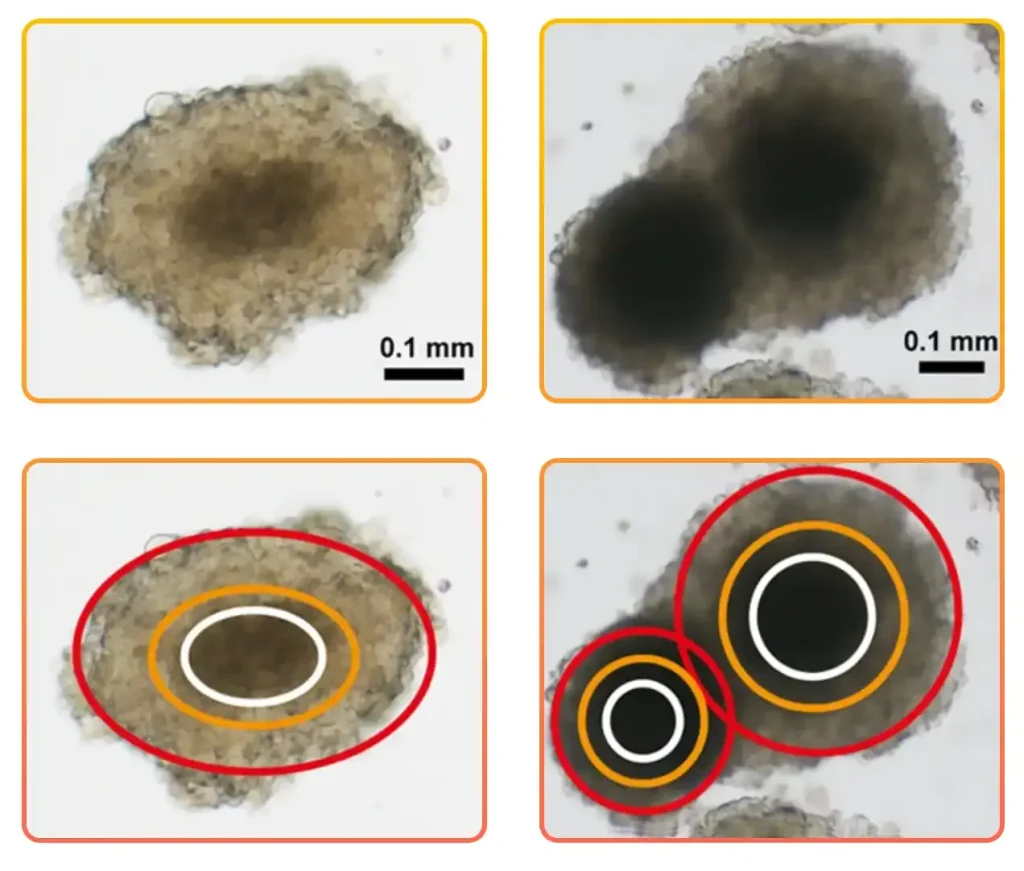
Figure 1. Variations in spheroid shape are also accompanied by changes in the dimension of the inner core and in the thickness of the external layers, mainly composed of actively proliferating cells. Brightfield images; scale bar = 0.1 mm. Adapted from: Zanoni M. et al. 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Sci Rep. 2016; 6:19103.
Zanoni et al. analyzed the effect of diverse spheroid parameters on drug response and concluded that the spherical shape is the most stable 3D structure. As an example, they observed that ellipsoidal spheroids showed substantial morphological changes due to cell detachment or budding of one or more small secondary spheroids. Spherically-shaped spheroids showed a more homogeneous distribution of the metabolic zones, reducing variability. The authors suggest selecting the most appropriate spherical colonies for use in cytotoxicity screening tests.
Parameters Influencing Uniform Spheroid Formation in 3D Cell Culture
Multiple variables influence how spheroids form and mature. Three broad categories stand out across the literature:
1. Fabrication Method
Spinner flasks, ultra-low attachment (ULA) plates, hanging drops, U-bottom microwells, and microfluidic droplets each create distinct microenvironments. Differences in shear forces, available surface area, and confinement strongly shape early aggregation and compaction. Studies show that spheroids derived from the same cell type but fabricated with different methods can diverge in size and morphology.
2. Intrinsic Cell Behavior
Cell proliferation rate, apoptosis, differentiation state, and ECM production all contribute to size variability. Some lines proliferate more intensely in 3D than in 2D due to enhanced cell-cell interactions, while others experience more pronounced hypoxia-induced quiescence or anoikis. These intrinsic mechanisms must be disentangled from fabrication-driven effects when interpreting drug responses.
3. Mass Transfer and Shear Stress
Larger spheroids face diffusion limitations that restrict the supply of oxygen and nutrients to their core. This affects not only viability but also gene expression and metabolic behavior. Dynamic culture systems introduce additional physical forces. Small eddies may shear aggregates, while large ones may impose benign mechanical stimulation. Uniform spheroid formation, therefore, requires controlled physical environments where shear and mass-transfer constraints do not introduce unwanted heterogeneity.
Comparing Methods for Scalable and Reproducible Spheroid Generation
No single spheroid formation assay is universally optimal. Instead, each offers trade-offs in throughput, uniformity, and operational complexity (Figure 2).
- Hanging Drop
A classical technique based on dispensing a small volume of cell suspension into a droplet on the lid of a plate, allowing gravity to pull the cells to the bottom of each droplet, where they aggregate. It is known for excellent uniformity, since each spheroid forms in an isolated droplet. However, it is low throughput, highly manual, and prone to evaporation due to the low volume of media used and media-exchange difficulties.
- Ultra-Low Attachment Plates and U-Bottom
This method relies on commercially available plates with a hydrophilic surface coating to prevent cell adhesion and maintain cells in suspension. It is simple, accessible, and scalable. Yet precise control of spheroid size is challenging unless cell concentration and handling steps are tightly optimized. For U-bottom plates, additional handling steps, such as pre-wetting, are often required to minimize bubble formation and maintain consistency.
- Spinner Flasks and Dynamic Culture Systems
Spinner flasks and bioreactors are dynamic culture systems that maintain cell suspension through continuous agitation, enabling scalable formation of three-dimensional aggregates. Spinner flasks promote spheroid formation through repeated cell collisions while preventing attachment to the vessel walls, allowing the production of large numbers of spheroids. However, they are difficult to standardize due to sensitivity to shear force and stirring conditions. In bioreactor-based systems, cell aggregation occurs randomly, often leading to spheroids or organoids with heterogeneous sizes, which can reduce experimental consistency in applications requiring reproducible 3D models.
- Microfluidic Droplet Systems
Microfluidic droplet systems generate spheroids by encapsulating cells inside uniform droplets made of an aqueous cell-culture phase dispersed within an immiscible oil phase. This method remains among the most precise approaches since droplets can encapsulate defined numbers of cells, leading to highly uniform spheroids with strong potential for automated workflows. Their complexity, the need for specialized chips, and the difficulty working with bigger organoids, though, limit adoption and their scale-up.

Figure 2. Comparison of five representative spheroid fabrication methods and their performance. Adapted from: Lee S, Lim J, Kwak B. Factors of bias in spheroid-based drug screening: Fabrication method, spheroid size, and cell viability. Biomed Pharmacother. 2025 Nov;192:118597.
Across these methods, a consistent theme emerges. High throughput and high uniformity rarely coexist without automation. Manual workflows introduce pipetting bias, inconsistent shear exposure, and variable operator technique. Even routine steps, such as medium exchange, can alter spheroid area, circularity, and viability when done manually. Precision liquid handling, imaging, and parameter logging are therefore essential components in achieving reproducible spheroid culture protocols at scale.
How to Ensure Quality Control and Traceability in High-Throughput Spheroid Workflows
Achieving uniformity is only half the challenge. Maintaining it throughout long-term assays requires systematic quality control, continuous monitoring, and robust traceability.
Automated imaging modules can verify morphology at each step and detect early deviations in size, shape, or structural integrity. Metadata logging is equally important. Recording environmental conditions, reagent lots, protocol versions, and timing ensures that subtle differences do not remain hidden sources of variability.
Platforms such as MO:BOT integrate these requirements by combining standardized liquid handling, gentle automated medium exchange, built-in imaging, and end-to-end data capture. In comparative studies, automated cultures maintained consistent spheroid morphology over multiple exchanges and demonstrated higher viability and lower coefficient of variation relative to manual workflows.
MO:BOT recognizes modules, tracks protocol execution, and generates fully traceable reports, ensuring that every sample in a high-throughput spheroid workflow shares the same fabrication conditions and culture history. The system automatically produces a detailed report at the end of each experiment, capturing experimental parameters and execution timelines. This level of standardization supports consistency between laboratories, facilitates regulatory submissions, and helps demonstrate the robustness and reliability of the in vitro model for its intended application. By reducing bias and enhancing reproducibility, MO:BOT enables teams to scale spheroid-based drug screening while maintaining scientific rigor.

If you’re working with spheroids and thinking about scale, let’s talk!
Sources
Decarli MC, Amaral R, Santos DPD, Tofani LB, Katayama E, Rezende RA, Silva JVLD, Swiech K, Suazo CAT, Mota C, Moroni L, Moraes ÂM. Cell spheroids as a versatile research platform: formation mechanisms, high throughput production, characterization and applications. Biofabrication. 2021 Apr 8;13(3). doi: 10.1088/1758-5090/abe6f2.
Lee S, Lim J, Kwak B. Factors of bias in spheroid-based drug screening: Fabrication method, spheroid size, and cell viability. Biomed Pharmacother. 2025 Nov;192:118597. doi: 10.1016/j.biopha.2025.118597.
Zanoni M, Piccinini F, Arienti C, Zamagni A, Santi S, Polico R, Bevilacqua A, Tesei A. 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Sci Rep. 2016; 6:19103.doi:10.1038/srep19103.
Blog Header image extracted from Heinrich MA, Huynh NT, Heinrich L, Prakash J. Understanding glioblastoma stromal barriers against NK cell attack using tri-culture 3D spheroid model. Heliyon. 2024 Jan 20;10(3):e24808. doi: 10.1016/j.heliyon.2024.e24808.